What we study (and why it matters)
Our research focus is cell division, a fundamental biological process. Cell division is complex with many opportunities for errors to occur. One potentially devastating outcome of errors in cell division is aneuploidy, which is defined by cells getting an incorrect number of chromosomes. Aneuploidy causes genetic disorders such as Down's Syndrome and has been implicated in tumorigenesis, tumor evolution and cancer metastasis.
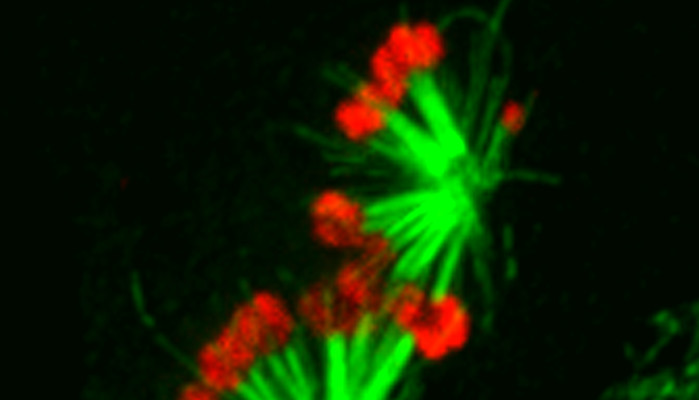
It is common for erroneous interactions between chromosomes and the spindle to occur, however, the cell possesses robust pathways to help combat these naturally occurring errors. The two major (and interconnected) pathways are the spindle assembly checkpoint (SAC), which delays anaphase onset in the presence of mal-oriented chromosomes, and the error correction pathway, which destabilizes erroneous interactions between chromosomal attachment sites called kinetochores (see below for more) and spindle microtubules. Aneuploidy ensues when the cell's surveillance and/or error correction systems fail. Our research aims to expand knowledge of how cells detect and correct errors during cell division. We address our central scientific questions with both molecular and biochemical approaches, high- and super-resolution microscopy of living cells, and plenty of fun.
Kinetochores (and more!)
A key player in the SAC is the kinetochore, a protein superstructure that assembles on each sister chromatid during cell division and consisting of nearly a hundred different proteins organized into a distinct and conserved molecular architecture.
The kinetochore's primary functions are two-fold. First, the kinetochore mediates the attachment of chromosomes to dynamic spindle microtubules, such that the chromosomes can be properly aligned and segregated. Faithful segregation requires that each chromosome becomes bioriented within the spindle, a configuration where each sister kinetochore is attached to microtubules emanating from opposing spindle poles. The second essential function of kinetochores is to initiate a "wait" signal that delays anaphase onset if any of the chromosomes are not bioriented.
We previously described a physical reorganization that takes place within the kinetochore, called intrakinetochore stretch, that correlates with kinetochore phosphorylation and generation of the "wait-anaphase" signal (Maresca & Salmon, JCB, 2009). The lab is actively investigating intrakinetochore stretch.
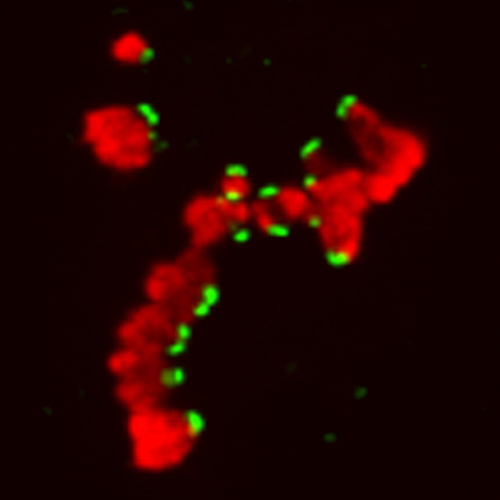
Syntelic attachments
In order to progress through cell division error-free, all cell must achieve biorientation, a state where all of the chromosomes are aligned such that each sister kinetochore is attached to opposing spindle poles. Unfortunately, kinetochores can improperly interact with spindle microtubules and, if not repaired, this often leads to chromosome mis-segregation and aneuploidy.

One common type of aberrant kinetochore-microtubule interaction is called a syntelic attachment, where both sister kinetochores are attached to the same pole. Syntelic attachments generate a "wait-anaphase" signal, so that the cell can delay in mitosis and work on converting syntelics to bioriented attachments. We have discovered that the application of elevated levels of force to chromosomes that are syntelically attached stabilizes these aberrant attachments and satisfies the SAC (Cane, Ye, et al., JCB, 2013).
We are using this live-cell force assay to further characterize syntelic attachments and how they are corrected. A central focus of the lab is to better understand how forces affect kinetochore-microtuble interactions and error correction.